技术交流
2.1 红锈,一种均匀腐蚀的形式
Rouging, a form of (uniform corrosion)
腐蚀基本上可以称为在一个部件表面不期望的、热力学引导的化学变化(在这种情况下:由不锈钢合金制成的)。
Corrosion can basically be referred to as an undesired, thermodynamically induced, chemical alteration of the surface of a component (in this case: one made of a stainless steel alloy).
在不锈钢合金技术中,已知由各种不同的因素和机制产生的更大范围的各种不同的局部和均匀的腐蚀效应,对该组件有或多或少严重的后果。
In stainless steel alloying technology, a wide range of different local and uniform corrosive effects, resulting from various different causes and mechanisms having more or less serious consequences for the component, is known.
就不锈钢表面的红锈效应来说,这种现象是由于60℃以上温度的缺氧超纯水、或超纯蒸汽的影响,富含铬氧化物钝化层的巨大变化、严重损坏甚至改变。这些变化随之会形成一个主要为氧化铁的红锈层,可以被部分擦除。上面描述的均匀腐蚀过程在相关的技术文献中被称为“红锈”,这是指通常可以被擦去的富含红色(富含氧化亚铁)物质颗粒的一个术语。
In the case of rouging effects on a stainless steel surface, the phenomenon is a massive alteration in, serious damage to, or even inversion of the chromium-oxide-rich passive layer due to the influence of oxygen-deficient ultrapure water at temperatures > 60 °C, or ultrapure steam. These changes then result in a predominantly iron oxide rouging layer, which can be partially wiped away. The uniform corrosion process described above is referred to as 'rouging' in the related technical literature, which is a term derived from the reddish (Feoxide-rich) material particles that can usually be wiped away.
从技术上讲,红锈描述了长期暴露在热、缺氧、去盐水下在不锈钢表面的典型的金黄色向红褐色薄层/变色形成。
Technically speaking, rouging describes the typical golden-yellow to reddish-brown formation of film/discolouration on stainless steel surfaces with long-term exposure to hot, oxygen-deficient, salt-free water.
在高温的清洁的蒸汽系统(T>100℃)中,甚至可以观察到附着在表面的红锈由深棕色到紫色膜的形式的过程。相关材料分析表明,在不锈钢表面形成的红锈膜主要由重金属氧化物颗粒(如铁、铬、镍等)组成,其中氧化铁含量明显占多数的。因此,可以肯定地假设,就它们的类型和与成分的关系而言,红锈沉积物都是特定不锈钢合金的腐蚀产物。
In clean-steam systems with high temperatures (T > 100 °C), it is even possible to observe rouging effects in the form of dark brown to violet films that normally stubbornly adhere to the surface. Relevant material analyses have shown that the rouge film formed on the stainless steel surface is essentially composed of heavy metal oxide particles (e.g. iron, chromium, nickel, etc.), with the iron oxide content clearly being predominant. As a result, it is safe to assume that—both with regard to their type and their relation to the composition—rouging deposits are a corrosion product of the specific stainless steel alloy.

图7:红锈层的形成[1]、[2](Formation of a rouge layer [1], [2])
因此,从热力学的角度来看,整个红锈过程背后的化学过程是不锈钢材料或它的金属原子的高级氧化状态,直接在与介质接触的表面上,这种状态远远超出了钝化反应。
From a thermodynamic standpoint, the che- mical process behind the entire rouging process is thus an advanced state of oxidation of the stainless steel material, or of its metal atoms, directly on the surface that has come into contact with media, a state that goes well beyond a passivation reaction.
结合缺氧纯水或清洁蒸汽作为环境条件质量为1.4301/1.4404/1.4435/1.4571等的奥氏体不锈钢表面,在高工艺温度(>60℃)对不锈钢表面的形态结构或自然再钝化性能有明显的负面影响(=钝化层的自我再生条件)。
In combination with pure, oxygen-deficient water or clean steam as environmental conditions for austenitic stainless steel surfaces in qualities of 1.4301/1.4404/1.4435/ 1.4571 etc., high process temperatures (> 60 °C) have clear negative effects on the morphological structure or natural repassivation property of stainless steel surfaces (= self-regeneration of the passive-layer conditions).
除了水中的缺氧外,高达100C的高水温也会导致水分子的额外分解(解离),并伴随着H+和oH离子的形成。由于氢氧化铁的形成量增加,这反过来又对钝化层的稳定性产生了负面的影响。与此同时,缺氧不仅导致去钝化,而且阻止任何进一步充分的再钝化(=铬的氧化),因此,去钝化和再钝化的动态平衡发生了关键的转变,这种平衡明显地向去钝化倾斜。
In addition to the oxygen depletion in the water, high water temperatures of up to 100 °C also cause an additional decompo- sition (dissociation) of water molecules accompanied by the formation of H+ and OH-ions. As a result of the increased formation of iron hydroxide, this in turn has a negative influence on the stability of the passive layer. At the same time, the oxygen deficiency causes not only a depassivation, but also prevents any further adequate repassivation (= oxidation of chromium), whereby a critical shift in the dynamic balance of deand repassivation occurs, with this balance clearly tipping towards depassivation.
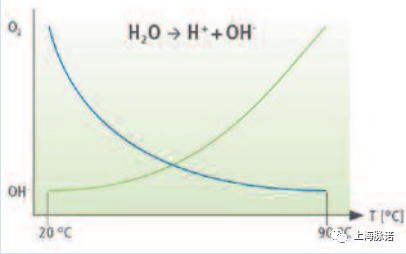
图8:水分子随温度升高的分解、蓝色曲线:含氧量降低、绿色曲线:水分解增加(离子形成)[1]。
Decomposition of water molecules as temperature increases Blue curve: Decrease in oxygen content Green curve: Increase in water decomposition (formation of ions) [1].
综上所述,这导致钝化层的有效性显著降低,并随着时间的推移,由于封闭的保护性氧化铬层的系统性破坏/分解,最终形成主要成分为铁氧化物的红锈层。在此过程中,以铁为主的基材的影响被放大。就可能性而言,这些变化本质上等于最初局部和最终完全的去钝化。
Taken together, this leads to a noticeable reduction in the passive layer's effectiveness and, over time, ultimately to the formation of the predominately iron oxide rouging layer, due to the systematic breakdown/decomposition of the closed protective chromium oxide layer. In the process, the impact of the iron-dominated base material is magnified. In terms of potential, these changes essentially amount to an initially local and eventually complete depassivation.
由于其键能,特别是在较高的温度下,铁显示对游离氢氧化根离子(水的)表现出更高的亲和力,这是温度导致的水分解增加的结果。在活化的不锈钢表面更多的氢氧化铁形式是作为二价铁和三价铁氧化物的前驱。
Due to its bonding energies, iron exhibits— particularly at higher temperatures—a higher affinity for the free hydroxide ions (of the water) that are present as a result of the temperature-induced increased decomposition of the water. More iron hydroxide forms on the activated stainless steel surface as a precursor of the iron(II) and iron(III) oxide.
在热力学引导的相反转中,主要的铬氧化物保护层,最初作为一个钝化表面存在,被转化为富含铁氧化物的层。这种新的铁氧化物层(=红锈层)形成了一个覆盖层,它有一个微孔的层状结构,这是典型的钢锈表面。红锈生产/形成背后的机制本质上是一种均匀腐蚀。在许多情况下,红锈的腐蚀产物与由于机理完全不同而造成的均匀腐蚀没有区别。
In a thermodynamically induced phase inversion, the predominantly chromium oxide protective layer, which was initially present as a passive surface, is converted into a layer rich in iron oxide. This new iron oxide layer (= rouging layer) forms a cover which has a microporous, layered structure that is typical of rusty steel surfaces. The mechanism behind the production/formation of rouging is essentially a form of uniform corrosion. In many cases, no distinction can be made between the corrosion products of rouging and the uniform corrosion due to completely different mechanisms.
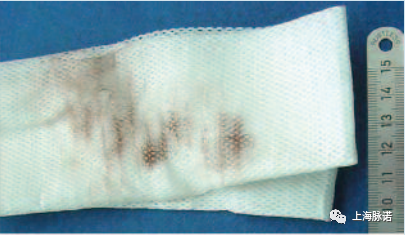
与其他类型的腐蚀相比,典型的红锈相对容易识别,通常是通过简单的拭子检测(见图9)。
Compared to other types of corrosion, typical rouging is relatively easy to identify, usually by means of a simple swab test (see Figure 9).
图9:红锈[6]覆盖的不锈钢表面的拭子样品(Swab sample from a stainless steel surface covered by rouging [6])
在许多应用情况下,特别是在蒸汽灭菌器中,由于部件表面出现典型的红锈膜由于水或水蒸气温度高缺氧和特定材料自身性能(=明确是不锈钢合金)(热力学)性能的相结合,无法避免伴随的显著变色。
In many application cases—especially in steam sterilizers—the occurrence of typical rouging films on the component surface, and the accompanying significant discolouration, cannot be avoided due to the combination of high water or water vapour temperatures, oxygen deficiency and the (thermodynamic) property of the specific material (= defined stainless steel alloy).
根据这些情况,运行人员需要指定预防和修复措施,以创造耐腐蚀的钝化表面环境。
In light of these circumstances, operators need to specify preventative and restorative measures in order to create corrosion-resistant passive surface conditions.


 微信公众号
微信公众号